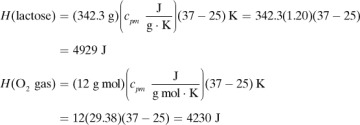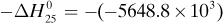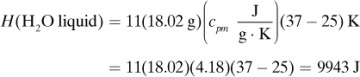- 1.0 Chapter Objectives
- 1.1 Classification of Transport Processes and Separation Processes (Unit Operations)
- 1.2 SI System of Basic Units Used in This Text and Other Systems
- 1.3 Methods of Expressing Temperatures and Compositions
- 1.4 Gas Laws and Vapor Pressure
- 1.5 Conservation of Mass and Material Balances
- 1.6 Energy and Heat Units
- 1.7 Conservation of Energy and Heat Balances
- 1.8 Numerical Methods for Integration
- 1.9 Chapter Summary
- Problems
- References
- Notation
1.7 Conservation of Energy and Heat Balances
1.7A Conservation of Energy
In making material balances, we used the law of conservation of mass, which states that the mass entering is equal to the mass leaving plus the mass left in the process. In a similar manner, we can state the law of conservation of energy, which states that all energy entering a process is equal to that leaving plus that left in the process. In this section, elementary heat balances will be made.
Energy can appear in many forms. Some of the common forms are enthalpy, electrical energy, chemical energy (in terms of ΔH reaction), kinetic energy, potential energy, work, and heat inflow.
In many cases in process engineering, which often takes place at constant pressure, electrical energy, kinetic energy, potential energy, and work either are not present or can be neglected. Then, only the enthalpy of the materials (at constant pressure), the standard chemical-reaction energy (ΔH0) at 25°C, and the heat added or removed must be taken into account in the energy balance. This is generally called a heat balance.
1.7B Heat Balances
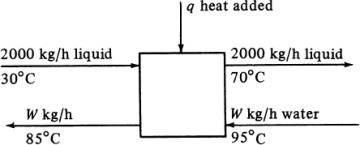
In making a heat balance at steady state, we use methods similar to those used in making a material balance. The energy or heat coming into a process in the inlet materials plus any net energy added to the process are equal to the energy leaving in the materials. Expressed mathematically,
where ΣHR is the sum of enthalpies of all materials entering the reaction process relative to the reference state for the standard heat of reaction at 298 K and 101.32 kPa. If the inlet temperature is above 298 K, this sum will be positive. 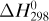 = standard heat of the reaction at 298 K and 101.32 kPa. The reaction contributes heat to the process, so the negative of
= standard heat of the reaction at 298 K and 101.32 kPa. The reaction contributes heat to the process, so the negative of 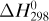 is taken to be positive input heat for an exothermic reaction. Also, q = net energy or heat added to the system. If heat leaves the system, this item will be negative. ΣHp = sum of enthalpies of all exiting materials referred to the standard reference state at 298 K (25°C).
is taken to be positive input heat for an exothermic reaction. Also, q = net energy or heat added to the system. If heat leaves the system, this item will be negative. ΣHp = sum of enthalpies of all exiting materials referred to the standard reference state at 298 K (25°C).
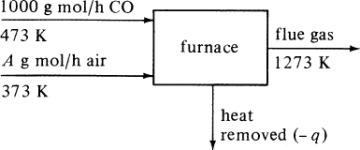
Note that if the materials coming into a process are below 298 K, ΣHR will be negative. Care must be taken not to confuse the signs of the items in Eq. (1.7-1). If no chemical reaction occurs, then simple heating, cooling, or phase change is occurring. Use of Eq. (1.7-1) will be illustrated by several examples. For convenience, it is common practice to call the terms on the left-hand side of Eq. (1.7-1) input items, and those on the right, output items.
Often, when chemical reactions occur in a process and the heat capacities vary with temperature, the solution in a heat balance can be trial and error if the final temperature is the unknown.
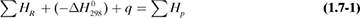

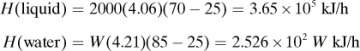
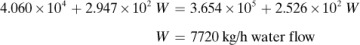
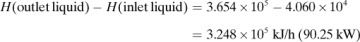
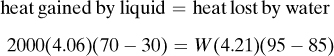
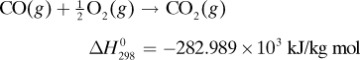


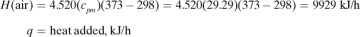
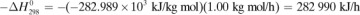
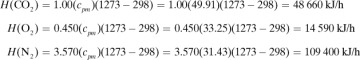
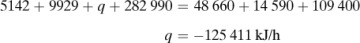
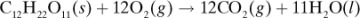
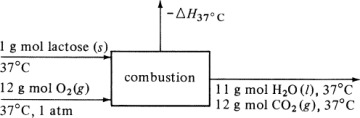
 in Appendix A.3 at 25°C is –5648.8 × 103 J/g mol. Calculate the heat of complete oxidation (combustion) at 37°C, which is the temperature of many biochemical reactions. The cpm of solid lactose is 1.20 J/g · K, and the molecular weight is 342.3 g mass/g mol.
in Appendix A.3 at 25°C is –5648.8 × 103 J/g mol. Calculate the heat of complete oxidation (combustion) at 37°C, which is the temperature of many biochemical reactions. The cpm of solid lactose is 1.20 J/g · K, and the molecular weight is 342.3 g mass/g mol.