- 9.0 Summary-Objectives
- 9.1 Introduction to Batch Distillation
- 9.2 Batch Distillation: Rayleigh Equation
- 9.3 Simple Binary Batch Distillation
- 9.4 Constant-Mole Batch Distillation
- 9.5 Batch Steam Distillation
- 9.6 Multistage Binary Batch Distillation
- 9.7 Multicomponent Simple Batch Distillation
- 9.8 Operating Time
- References
- Homework
- Chapter 9 Appendix A. Spreadsheet for Simple Multicomponent Batch Distillation, Constant Relative Volatility
9.1 Introduction to Batch Distillation
Continuous distillation is a thermodynamically efficient method of producing large amounts of material of constant composition. When small amounts of material or varying product compositions are required, batch distillation has several advantages. In batch distillation a charge of feed is loaded into the reboiler, the steam is turned on, and after a short startup period, product can be withdrawn from the top of the column. When the distillation is finished, the heat is shut off, and the material left in the reboiler is removed. Then a new batch can be started. Usually the distillate is the desired product.
Batch distillation is a much older process than continuous distillation. Mesopotamian clay distillation pots have been dated to around 3500 B.C.E. (RT, 2007), and alchemists in Alexandria used simple batch retorts in the first century A.D. (Davies, 1980). Batch distillation was developed to concentrate alcohol by Arab alchemists around 700 A.D. (Vallee, 1998). It was adopted in Western Europe, and the first known book on the subject was Hieronymus Brunschwig’s Liber de arte distillandi, published in Latin in the early 1500s. This book remained a standard pharmaceutical and medical text for more than a century. The first distillation book written for a literate but not the scholarly community was Walter Ryff’s Das New gross Distillier Buch, published in German in 1545 (Stanwood, 2005). This book included a “listing of distilling apparatus, techniques, and the plants, animals, and minerals able to be distilled for human pharmaceutical use.” Kockmann (2014) thoroughly reviews the history of batch distillation. Advances in batch distillation have been associated with its use to distil alcohol, pharmaceuticals, coal oil, petroleum oil, and fine chemicals.
Batch distillation is versatile. A run may last from a few hours to several days. Batch distillation is used when the plant does not run continuously and batches must be completed in one or two shifts (8 to 16 h). It is often used when the same equipment distills several different products at different times. If distillation is required only occasionally, batch distillation is the usual choice.
Equipment can be arranged in a variety of configurations (Sorensen, 2014). In simple batch distillation (Figure 9-1), the vapor is withdrawn continuously from the reboiler. The system differs from flash distillation in that there is no continuous feed input, and the liquid is drained only at the end of the batch. A similar alternative is constant-mole batch distillation in which solvent is fed continually to the still pot to keep the number of moles constant (see Section 9-4).
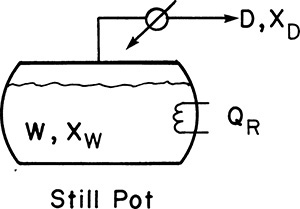
FIGURE 9-1 Simple batch distillation
In a multistage batch distillation, a staged or packed column is placed above the reboiler as in Figure 9-2. Reflux is returned to the column. In the usual operation, distillate is withdrawn continually (Diwekar, 1995; Luyben, 1971; Mujtaba, 2004; Pratt, 1967; Robinson and Gilliland, 1950; Sorensen, 2014) until the column is shut down and drained. In an alternative method (Treybal, 1970), no distillate is withdrawn; instead, the composition of liquid in the accumulator changes. When the distillate in the accumulator is of the desired composition and in the desired amount, both the accumulator and the reboiler are drained. Luyben (1971) indicated that the usual method should be superior; however, the alternative method may be simpler to operate.
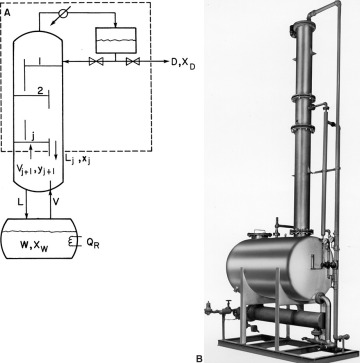
FIGURE 9-2 Multistage batch distillation; (A) schematic, (B) photograph of packaged batch distillation/solvent recovery system of approximately 400-gallon capacity. Courtesy of APV Equipment, Inc., Tonowanda, New York
Another alternative is called inverted batch distillation (Diwekar, 1995; Pratt, 1967; Robinson and Gilliland, 1950) because bottoms are withdrawn continuously while distillate is withdrawn only at the end of the distillation (see Figure 9-8 and Problems 9.C2 and 9.E3). In this case the charge is placed in the accumulator, and a reboiler with a small holdup is used. Inverted batch distillation is seldom used, but it is useful when quite pure bottoms product is required. Additional configurations include middle-vessel, multivessel, extractive, and hybrid (Sorensen, 2014).
