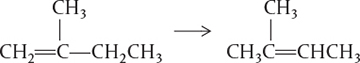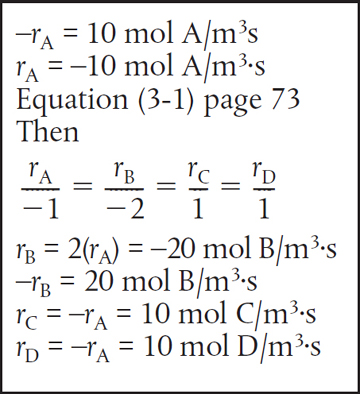Mole Balances
Mole balances are the first building block of chemical reaction engineering. In this excerpt, learn how to define the rate of reaction, apply the mole balance equation to the four most common types of industrial reactors, and more.
Save 35% off the list price* of the related book or multi-format eBook (EPUB + MOBI + PDF) with discount code ARTICLE.
* See informit.com/terms
The first step to knowledge is to know that we are ignorant.
—Socrates (470-399 B.C.)
The Wide Wild World of Chemical Reaction Engineering
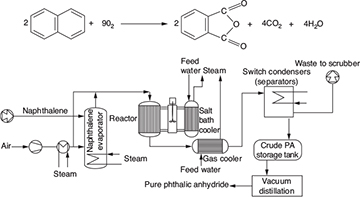
Chemical kinetics is the study of chemical reaction rates and reaction mechanisms. The study of chemical reaction engineering (CRE) combines the study of chemical kinetics with the reactors in which the reactions occur. Chemical kinetics and reactor design are at the heart of producing almost all industrial chemicals, such as the manufacture of phthalic anhydride shown in Figure 1-1. It is primarily a knowledge of chemical kinetics and reactor design that distinguishes the chemical engineer from other engineers. The selection of a reaction system that operates in the safest and most efficient manner can be the key to the economic success or failure of a chemical plant. For example, if a reaction system produces a large amount of undesirable product, subsequent purification and separation of the desired product could make the entire process economically unfeasible.
Figure 1-1 Manufacture of phthalic anhydride.
The chemical reaction engineering (CRE) principles learned here can also be applied in many areas, such as waste treatment, microelectronics, nanoparticles, and living systems, in addition to the more traditional areas of the manufacture of chemicals and pharmaceuticals. Some of the examples that illustrate the wide application of CRE principles in this book are shown in Figure 1-2. These examples include modeling smog in the Los Angeles (L.A.) basin (Chapter 1), the digestive system of a hippopotamus (Chapter 2 on the CRE Web site, www.umich.edu/~elements/5e/index.html), and molecular CRE (Chapter 3). Also shown are the manufacture of ethylene glycol (antifreeze), where three of the most common types of industrial reactors are used (Chapters 5 and 6), and the use of wetlands to degrade toxic chemicals (Chapter 7 on the CRE Web site). Other examples shown are the solid-liquid kinetics of acid-rock interactions to improve oil recovery (Chapter 7); pharmacokinetics of cobra bites (Chapter 8 Web Module); free-radical scavengers used in the design of motor oils (Chapter 9); enzyme kinetics (Chapter 9) and drug delivery pharmacokinetics (Chapter 9> on the CRE Web site); heat effects, runaway reactions, and plant safety (Chapters 11 through 13); and increasing the octane number of gasoline and the manufacture of computer chips (Chapter 10).
Figure 1-2 The wide world of CRE applications.
1.1 The Rate of Reaction, –rA
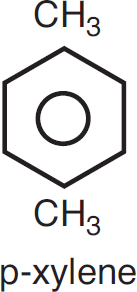
The rate of reaction tells us how fast a number of moles of one chemical species are being consumed to form another chemical species. The term chemical species refers to any chemical component or element with a given identity. The identity of a chemical species is determined by the kind, number, and configuration ofthat species’ atoms. For example, the species para-xylene is made up of a fixed number of specific atoms in a definite molecular arrangement or configuration. The structure shown illustrates the kind, number, and configuration of atoms on a molecular level. Even though two chemical compounds have exactly the same kind and number of atoms of each element, they could still be different species because of different configurations. For example, 2-butene has four carbon atoms and eight hydrogen atoms; however, the atoms in this compound can form two different arrangements.
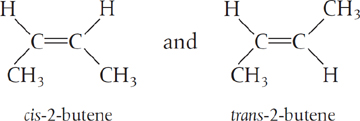
As a consequence of the different configurations, these two isomers display different chemical and physical properties. Therefore, we consider them as two different species, even though each has the same number of atoms of each element.
We say that a chemical reaction has taken place when a detectable number of molecules of one or more species have lost their identity and assumed a new form by a change in the kind or number of atoms in the compound and/or by a change in structure or configuration of these atoms. In this classical approach to chemical change, it is assumed that the total mass is neither created nor destroyed when a chemical reaction occurs. The mass referred to is the total collective mass of all the different species in the system. However, when considering the individual species involved in a particular reaction, we do speak of the rate of disappearance of mass of a particular species. The rate of disappearance of a species, say species A, is the number of A molecules that lose their chemical identity per unit time per unit volume through the breaking and subsequent re-forming of chemical bonds during the course of the reaction. In order for a particular species to “appear” in the system, some prescribed fraction of another species must lose its chemical identity.
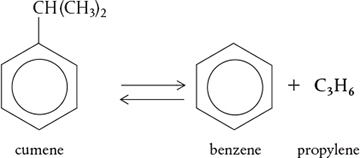
There are three basic ways a species may lose its chemical identity: decomposition, combination, and isomerization. In decomposition, the molecule loses its identity by being broken down into smaller molecules, atoms, or atom fragments. For example, if benzene and propylene are formed from a cumene molecule,
the cumene molecule has lost its identity (i.e., disappeared) by breaking its bonds to form these molecules. A second way that a molecule may lose its chemical identity is through combination with another molecule or atom. In the above reaction, the propylene molecule would lose its chemical identity if the reaction were carried out in the reverse direction, so that it combined with benzene to form cumene. The third way a species may lose its chemical identity is through isomerization, such as the reaction
Here, although the molecule neither adds other molecules to itself nor breaks into smaller molecules, it still loses its identity through a change in configuration.
To summarize this point, we say that a given number of molecules (i.e., moles) of a particular chemical species have reacted or disappeared when the molecules have lost their chemical identity.
The rate at which a given chemical reaction proceeds can be expressed in several ways. To illustrate, consider the reaction of chlorobenzene with chloral to produce the banned insecticide DDT (dichlorodiphenyl-trichloroethane) in the presence of fuming sulfuric acid.
CCl3CHO + 2C6H5Cl → (C6H4Cl)2CHCC13 + H2O
Letting the symbol A represent chloral, B be chlorobenzene, C be DDT, and D be H2O, we obtain
A + 2B → C + D
The numerical value of the rate of disappearance of reactant A, −rA, is a positive number.
The rate of reaction, −rA, is the number of moles of A (e.g., chloral) reacting (disappearing) per unit time per unit volume (mol/dm3·s).
Example 1-1 Rates of Disappearance and Formation
Chloral is being consumed at a rate of 10 moles per second per m3 when reacting with chlorobenzene to form DDT and water in the reaction described above. In symbol form, the reaction is written as
A + 2B → C + D
Write the rates of disappearance and formation (i.e., generation) for each species in this reaction.
Solution
(a) Chloral[A]: |
The rate of reaction of chloral [A] (—rA) is given as 10mol/m3·s Rate of disappearance of A = –rA = 10 mol/m3·s Rate of formation of A = rA = –10 mol/m3·s |
(b) Chlorobenzene[B]: |
For every mole of chloral that disappears, two moles of chlorobenzene [B] also disappear. Rate of disappearance of B = –rB = 20 mol/m3·s Rate of formation of B = rB = –20 mol/m3·s |
(c) DDT[C]: |
For every mole of chloral that disappears, two moles of DDT[C] appears. Rate of formation of C = rC = 10 mol/m3·s Rate of disappearance of C = –rC = –10 mol/m3·s |
(d) Water[D]: |
Same relationship to chloral as the relationship to DDT Rate of formation of D = rD = 10 mol/m3·s Rate of disappearance of D = –rD = –10 mol/m3·s |
Analysis: The purpose of this example is to better understand the convention for the rate of reaction. The symbol rj is the rate of formation (generation) of species j. If species j is a reactant, the numerical value of rj will be a negative number. If species j is a product, then rj will be a positive number. The rate of reaction, −rA, is the rate of disappearance of reactant A and must be a positive number. A mnemonic relationship to help remember how to obtain relative rates of reaction of A to B, etc., is given by Equation (3-1) on page 73.
In Chapter 3, we will delineate the prescribed relationship between the rate of formation of one species, r> (e.g., DDT [C]), and the rate of disappearance of another species, - η (e.g., chlorobenzene [B]), in a chemical reaction.
Heterogeneous reactions involve more than one phase. In heterogeneous reaction systems, the rate of reaction is usually expressed in measures other than volume, such as reaction surface area or catalyst weight. For a gas-solid catalytic reaction, the gas molecules must interact with the solid catalyst surface for the reaction to take place, as described in Chapter 10.
The dimensions of this heterogeneous reaction rate,
Most of the introductory discussions on chemical reaction engineering in this book focus on homogeneous systems, in which case we simply say that η is the rate of formation of species j per unit volume. It is the number of moles of species j generated per unit volume per unit time.
We can say four things about the reaction rate rj. The reaction rate law for rj is
The rate of formation of species; (mole/time/volume)
An algebraic equation
Independent of the type of reactor (e.g., batch or continuous flow) in which the reaction is carried out
Solely a function of the properties of the reacting materials and reaction conditions (e.g., species concentration, temperature, pressure, or type of catalyst, if any) at a point in the system
However, because the properties and reaction conditions of the reacting materials may vary with position in a chemical reactor, rj can in turn be a function of position and can vary from point to point in the system.
The chemical reaction rate law is essentially an algebraic equation involving concentration, not a differential equation.1 For example, the algebraic form of the rate law for −rA for the reaction
A → products
may be a linear function of concentration,
or it may be some other algebraic function of concentration, such as Equation 3-6 shown in Chapter 3
or
For a given reaction, the particular concentration dependence that the rate law follows (i.e., −rA = kCA or