1.6 Summary
Years from now you may have some difficulty recalling the details presented in this text. On the other hand, the two basic premises outlined in this introductory section are so fundamental to technically educated people that you really should commit them to long-term memory as soon as possible. Formally, we may state our two basic premises as the first and second "laws" of thermodynamics.13
- First Law: Overall energy is conserved (you can't get something for nothing).
- Second Law: Overall entropy changes are greater than or equal to zero (generation of disorder results in lost work).
The first law is further developed in Chapters 2 and 3. The concepts of entropy and the second law are developed in Chapter 4 and process applications in Chapter 5. The exact relationship between the two basic premises and these two laws may not become apparent until some time later in this text, but you should begin to absorb and contemplate these fundamentals now. There are times when the endeavor to apply these simple laws seems daunting, but the answer appears simple in retrospect, once obtained. By practicing adaptation of the basic principles to many specific problems, you slowly grasp the appropriate connection between the basic premises and finding the details. Try not to be distracted by the vocabulary or the tedious notation that goes into keeping all the coupled systems classified in textbook fashion. Keep in mind that other students have passed through this and found the detailed analysis to be worth the effort.
Important Equations
The content of this chapter is primarily about laying down the fundamental concepts more than deriving equations. Nevertheless, three concepts that we refer to repeatedly can be expressed by equations. Of course, the ideal gas law is important, but an implication of it that may be new is:
Equation 1.28
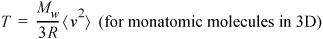
This equation conveys that temperature is closely related to molecular kinetic energy. Although derived with the ideal gas assumption, it turns out to be true for real fluids as well. Every time you alter the temperature, you should think about the implications for molecular kinetic energy. Another important equation relates to deviations from the ideal gas law:
Equation 1.29
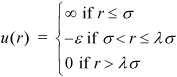
This is the square well potential model, the simplest characterization of how real molecules attract and repel. As you add energy to the system, real fluids may absorb that energy by moving molecules from inside the square well to outside, converting potential energy into kinetic energy without altering the temperature as an ideal gas would. A simple example is boiling. Ideal gases cannot boil, but real fluids can. This interplay between kinetic energy, temperature, and potential energy pervades many discussions throughout the text.
Finally, we can write a generic equation that symbolizes the procedure for interpolation:
Equation 1.30

A similar equation is used for quality calculations which can be viewed as an interpolation between saturated liquid and saturated vapor. Throughout Unit I, we refer extensively to the steam tables and interpolation to account for deviations from the ideal gas law.
Test Yourself
- Draw a sketch of the force model implied by the square-well potential, indicating the position(s) where the force between two atoms is zero and the positions where it is nonzero.
- Explain in words how the pressure of a fluid against the walls of its container is related to the velocity of the molecules.
- What is it about molecules that requires us to add heat to convert liquids to gases?
- If the kinetic energy of pure liquid and vapor molecules at phase equilibrium must be the same, and the internal energy of a system is the sum of the kinetic and potential energies, what does this say about the intensive internal energy of a liquid phase compared with the intensive internal energy of the gas phase?
- Explain the terms "energy," "potential energy," "kinetic energy," and "internal energy."
- How is the internal energy of a substance related to the intermolecular pair potentials of the molecules?
- Are T and P intensive properties? Name two intensive properties and two extensive properties.
- How many degrees of freedom exist when a pure substance coexists as a liquid and gas?
- Can an ideal gas condense? Can real fluids that follow the ideal gas law condense?
- Give examples of bubble, dew, saturation, and superheated conditions. Explain what is meant when wet steam has a quality of 25%.
- Create and solve a problem that requires double interpolation.
