- 1.1 Fluid Mechanics in Chemical Engineering
- 1.2 General Concepts of a Fluid
- 1.3 Stresses, Pressure, Velocity, and the Basic Laws
- 1.4 Physical Properties-Density, Viscosity, and Surface Tension
- 1.5 Units and Systems of Units
- 1.6 Hydrostatics
- 1.7 Pressure Change Caused by Rotation
- Problems for Chapter 1
1.2 General Concepts of a Fluid
We must begin by responding to the question, “What is a fluid?” Broadly speaking, a fluid is a substance that will deform continuously when it is subjected to a tangential or shear force, much as a similar type of force is exerted when a water-skier skims over the surface of a lake or butter is spread on a slice of bread. The rate at which the fluid deforms continuously depends not only on the magnitude of the applied force but also on a property of the fluid called its viscosity or resistance to deformation and flow. Solids will also deform when sheared, but a position of equilibrium is soon reached in which elastic forces induced by the deformation of the solid exactly counterbalance the applied shear force, and further deformation ceases.
A simple apparatus for shearing a fluid is shown in Fig. 1.1. The fluid is contained between two concentric cylinders; the outer cylinder is stationary, and the inner one (of radius R) is rotated steadily with an angular velocity ω. This shearing motion of a fluid can continue indefinitely, provided that a source of energy—supplied by means of a torque here—is available for rotating the inner cylinder. The diagram also shows the resulting velocity profile; note that the velocity in the direction of rotation varies from the peripheral velocity Rω of the inner cylinder down to zero at the outer stationary cylinder, these representing typical no-slip conditions at both locations. However, if the intervening space is filled with a solid—even one with obvious elasticity, such as rubber—only a limited rotation will be possible before a position of equilibrium is reached, unless, of course, the torque is so high that slip occurs between the rubber and the cylinder.
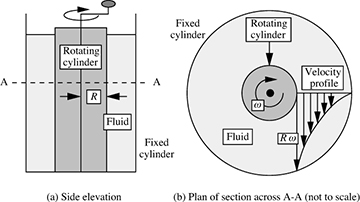
Fig. 1.1 Shearing of a fluid.
There are various classes of fluids. Those that behave according to nice and obvious simple laws, such as water, oil, and air, are generally called Newtonian fluids. These fluids exhibit constant viscosity but, under typical processing conditions, virtually no elasticity. Fortunately, a very large number of fluids of interest to the chemical engineer exhibit Newtonian behavior, which will be assumed throughout the book, except in Chapter 11, which is devoted to the study of non-Newtonian fluids.
A fluid whose viscosity is not constant (but depends, for example, on the intensity to which it is being sheared), or which exhibits significant elasticity, is termed non-Newtonian. For example, several polymeric materials subject to deformation can “remember” their recent molecular configurations, and in attempting to recover their recent states, they will exhibit elasticity in addition to viscosity. Other fluids, such as drilling mud and toothpaste, behave essentially as solids and will not flow when subject to small shear forces, but will flow readily under the influence of high shear forces.
Fluids can also be broadly classified into two main categories—liquids and gases. Liquids are characterized by relatively high densities and viscosities, with molecules close together; their volumes tend to remain constant, roughly independent of pressure, temperature, or the size of the vessels containing them. Gases, on the other hand, have relatively low densities and viscosities, with molecules far apart; generally, they will compleetely fill the container in which they are placed. However, these two states—liquid and gaseous—represent but the two extreme ends of a continuous spectrum of possibilities.
The situation is readily illustrated by considering a fluid that is initially a gas at point G on the pressure/temperature diagram shown in Fig. 1.2. By increasing the pressure, and perhaps lowering the temperature, the vapor-pressure curve is soon reached and crossed, and the fluid condenses and apparently becomes a liquid at point L. By continuously adjusting the pressure and temperature so that the clockwise path is followed, and circumnavigating the critical point C in the process, the fluid is returned to G, where it is presumably once more a gas. But where does the transition from liquid at L to gas at G occur? The answer is at no single point, but rather that the change is a continuous and gradual one, through a whole spectrum of intermediate states.
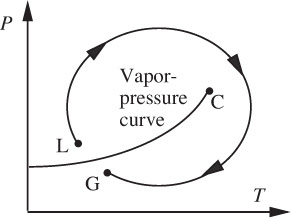
Fig. 1.2 When does a liquid become a gas?
