Problems
- The IPAT equation Use the IPAT equation to estimate the percentage increase in the amount of energy that would be required, worldwide, in 2050, relative to 2006. To estimate the increase in population and affluence (the P and A in the IPAT equation), assume that population grows 1% per year and that global economic activity per person grows 2% per year. Assume that the energy consumption per dollar of GDP (the T in the IPAT equation) remains at 2006 levels. How much does this estimate change if population growth is 2% and economic growth is 4%?
- Affluence and energy use Estimate the amount of energy that will be used annually, worldwide, if over the next 50 years world population grows to 10 billion and energy use per capita increases to the current per capita consumption rate in the United States. What percentage increase does this represent over current global energy use?
- Energy efficiency in automobiles Assume that the conversion of energy into mechanical work (at the wheel) in an internal combustion engine is 20%. Calculate the gallons of gasoline required to deliver 30 horsepower at the wheel, for one hour.
- Water use by automobiles Assuming that generating a kilowatt-hour of electricity requires an average of 13 gal of water (Example 1-3) and that an average electric vehicle requires 0.3 kWh/mi traveled (Kintner-Meyer et al., 2007), calculate the water use per mile traveled for an electric vehicle. If gasoline production requires approximately 10 gallons of water per gallon produced and an average gasoline-powered vehicle has a fuel efficiency of 25 mpg, calculate the water use per mile traveled of a gasoline-powered vehicle.
- Energy efficiency in lighting Assume that a 25-watt fluorescent bulb provides the same illumination as a 100-watt incandescent bulb. Calculate the mass of coal that would be required, over the 8000-hour life of the fluorescent bulb, to generate the additional electricity required for an incandescent bulb. Assume transmission losses of 10%, 40% efficiency of electricity generation, and 10,000 BTU/lb for the heat of combustion of coal.
- Energy savings potential of compact fluorescent versus incandescent lightbulbs Compact fluorescent lightbulbs provide similar lighting characteristics to incandescent bulbs yet use just one-fourth of the energy. Estimate the energy savings potential on a national scale of replacing all incandescent bulbs in home (residential) lighting applications with compact fluorescent bulbs. In 2008, total U.S. energy consumption was 99.3 quadrillion (1015) BTU (quads), and electricity in all applications consumed 40.1 quads of primary energy. Assume that residential lighting is 3% of all electricity consumption in the United States and that all energy consumption for residential lighting is due to incandescent bulb use. How large (%) is the energy savings compared to annual U.S. energy consumption (2008 reference year)? Is this savings significant?
- Global energy balance: no atmosphere (adapted from Wallace and Hobbs, 1977)
- The figure below is a schematic diagram of the Earth in radiative equilibrium with its surroundings, assuming no atmosphere. Radiative equilibrium requires that the rate of radiant (solar) energy absorbed by the surface must equal the rate of radiant energy emitted (infrared). Let S be the incident solar irradiance (1360 W/m2), E the infrared planetary irradiance (W/m2), R E the radius of the Earth (meters), and A the planetary albedo (0.3). The albedo is the fraction of total incident solar radiation reflected back into space without being absorbed.
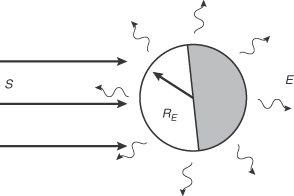
- Write the steady-state energy balance equation, assuming radiative equilibrium as stated above. Solve for the infrared irradiance, E, and show that its value is 238 W/m2.
- Solve for the global average surface temperature (K), assuming that the surface emits infrared radiation as a black body. In this case, the Stefan-Boltzman Law for a black body is E = σ T4 s is the Stefan-Boltzman constant (5.67 × 10–8 W/(m2 ·˚K4)), and T is absolute temperature (˚K). Compare this temperature with the observed global average surface temperature of 280 K. Discuss possible reasons for the difference.
- The figure below is a schematic diagram of the Earth in radiative equilibrium with its surroundings, assuming no atmosphere. Radiative equilibrium requires that the rate of radiant (solar) energy absorbed by the surface must equal the rate of radiant energy emitted (infrared). Let S be the incident solar irradiance (1360 W/m2), E the infrared planetary irradiance (W/m2), R E the radius of the Earth (meters), and A the planetary albedo (0.3). The albedo is the fraction of total incident solar radiation reflected back into space without being absorbed.
- Global energy balance: with a greenhouse gas atmosphere (adapted from Wallace and Hobbs, 1977) Refer to the schematic diagram below for energy balance calculations on the atmosphere and surface of the Earth. Assume that the atmosphere can be regarded as a thin layer with an absorbtivity of 0.1 for solar radiation and 0.8 for infrared radiation. Assume that the Earth’s surface radiates as a black body (absorbtivity = emissivity = 1.0).
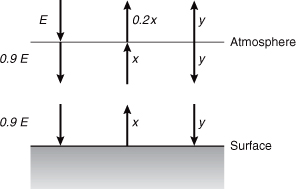
Let x equal the irradiance (W/m2) of the Earth’s surface and y the irradiance (both upward and downward) of the atmosphere. E is the irradiance entering the Earthatmosphere system from space averaged over the globe (E = 238 W/m2 from Problem 7). At the Earth’s surface, a radiation balance requires that
0.9E + y = x
(irradiance in = irradiance out)while for the atmosphere layer, the radiation balance is
E + x = 0.9E + 2y + 0.2x
- Solve these equations simultaneously for y and x.
- Use the Stefan-Boltzman Law (see Problem 7) to calculate the temperatures of both the surface and the atmosphere. Show that the surface temperature is higher than when no atmosphere is present (Problem 7).
- The emission into the atmosphere of infrared-absorbing chemicals is a concern for global warming. Determine by how much the absorbtivity of the atmosphere for infrared radiation must increase in order to cause a rise in the global average temperature by 1°C above the value calculated in Part b.
Global carbon dioxide mass balance Recent estimates of carbon dioxide emission rates to and removal rates from the atmosphere result in the following schematic diagram (EIA, 1998):
The numbers in the diagram have units of 109 metric tons of carbon per year, where a metric ton is equal to 1000 kg. To calculate the emission and removal rates for carbon dioxide, multiply each number by the ratio of molecular weights (44 g CO2/12 g C).
- Write a steady-state mass balance for carbon dioxide in the atmosphere and calculate the rate of accumulation of CO2 in the atmosphere in units of kilograms per year. Is the accumulation rate positive or negative?
- Change the emission rate due to fossil fuel combustion by +10% and recalculate the rate of accumulation of CO2 in the atmosphere in units of kilograms per year. Compare this to the change in the rate of accumulation of CO2 in the atmosphere due to a +1% change in carbon dioxide release by microorganisms.
- Calculate the rate of change in CO2 concentration in units of parts per million per year, and compare this number with the observed rate of change stated in Section 1.3.2. Recall the definition of parts per million (ppm), which for CO2 is the mole fraction of CO2 in the air. Assume that we are considering only the first 10 km in height of the atmosphere and that its gases are well mixed. Take for this calculation that the total moles of gas in the first 10 km of the atmosphere is approximately 1.5 × 1020.
- Describe how the rate of accumulation of CO2 in the atmosphere, calculated in Parts b and c, would change if processes such as carbon dioxide fertilization and forest growth increase as CO2 concentrations increase. What processes releasing CO2 might increase as atmospheric concentrations increase? (Hint: Assume that temperature will rise as CO2 concentrations rise.)
- Electric vehicles: effects on industrial production of fuels Replacing automobiles having internal combustion engines with vehicles having electric motors is seen by some as the best solution to urban smog and tropospheric ozone. Write a short report (one to two pages double-spaced) on the likely effects of this transition on industrial production of fuels. Assume for this analysis that the amount of energy required per mile traveled is roughly the same for each kind of vehicle. Consider the environmental impacts of using different kinds of fuel for the electricity generation to satisfy the demand from electric vehicles. Background reading for this problem is found in Graedel and Allenby (1998).
- Essay on an environmental issue Read an article from a science or engineering journal, from a popular magazine, or from the Internet on some environmental issue that is of interest to you. Summarize the article in a short memorandum format, addressed to your instructor. In the body of the memorandum, limit the length to one page of single-spaced text, including graphics/tables (if needed). Structure the memorandum in this way:
- Introduction and motivation
- A description of the issue
- A description of what engineers are doing, have done, or are going to do to address the challenge
Use of headings is appropriate, and be sure to reference information sources.
Potential topics:
- Stratospheric ozone depletion: the chemical industry connection
- Smog in industrialized urban areas
- Toxic chemicals in commerce and in the environment
- Industrial hazardous waste generation and management
- Environmental challenges for genetically engineered foods
- The cleanup of industrial sites (Superfund program)
- Pollution prevention issues, technologies, or initiatives
- Endocrine disruptors: What are they, why are they harmful, and what is the chemical industry doing about them?
- Environmental effects (advantages/disadvantages) of biodiesel or corn/cellulosic ethanol for transportation fuels
- Fuel cells and their environmental consequences
- Water resources: quality and quantity
- Petroleum: Are we running out? What are the alternatives?
- Renewable energy: What is it, and can it make a difference?
Potential sources of information:
Scientific and engineering research journals (check the library current journals section):
- Environmental Science & Technology
- Environmental Progress and Sustainable Energy
- Industrial and Engineering Chemistry Research
- Chemical and Engineering News
- Science
- Scientific American
Internet resources:
- American Chemistry Council (formerly the Chemical Manufacturers Association)
- U.S. Environmental Protection Agency (www.epa.gov)
- Your state’s Department of Environmental Quality
- Sustainable development An overview of the Report of the World Commission on Environment and Development is at www.un-documents.net/ocf-ov.htm. A number of global challenges to the environment, economic development, and living conditions were discussed. Summarize one or two of the key challenges in a memo format in one or two pages.
- International trade in waste One of the consequences of the globalization of trade is a growing global trade in waste, often toxic and hazardous waste. Read a recent article on this subject and write a one-to two-page memo on key findings. Include a reference or references in your memo. One possible source of information is the International Network for Environmental Compliance and Enforcement (www.inece.org/seaport/SeaportWorkingPaper_24November.pdf).

